Quick Guide to assembling a dental tray [AS4187 product families]
 The latest version of AS/NZS 4187 released on the 15th December 2014 brought some very significant changes to the previous standard. One of the most significant changes is the standards reference to Product Families.
The latest version of AS/NZS 4187 released on the 15th December 2014 brought some very significant changes to the previous standard. One of the most significant changes is the standards reference to Product Families.
In my blog "Here's how to make sense of the AS4187 categorisation of instrument product families", I provided an overview of ISO/TS 17665-3:2013 - Sterilization of health care products - Moist heat - Part 3: Guidance on the designation of a medical device to a product family and processing category for steam sterilization.
In today's post I will get into the specifics of how to apply the standard to a dental assessment tray.
Dental Tray Assembly Table
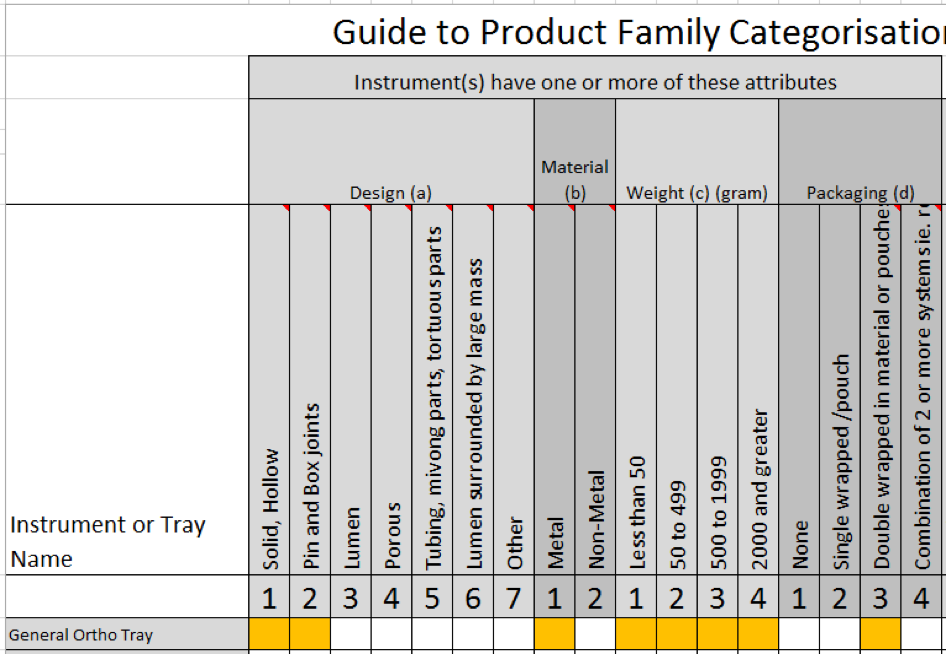
The diagram below shows the makeup of a typical a Dental Assessment Tray. Now, using our downloadable ‘Guide to Product Family Categorisation’, we will set the attributes for each instrument.
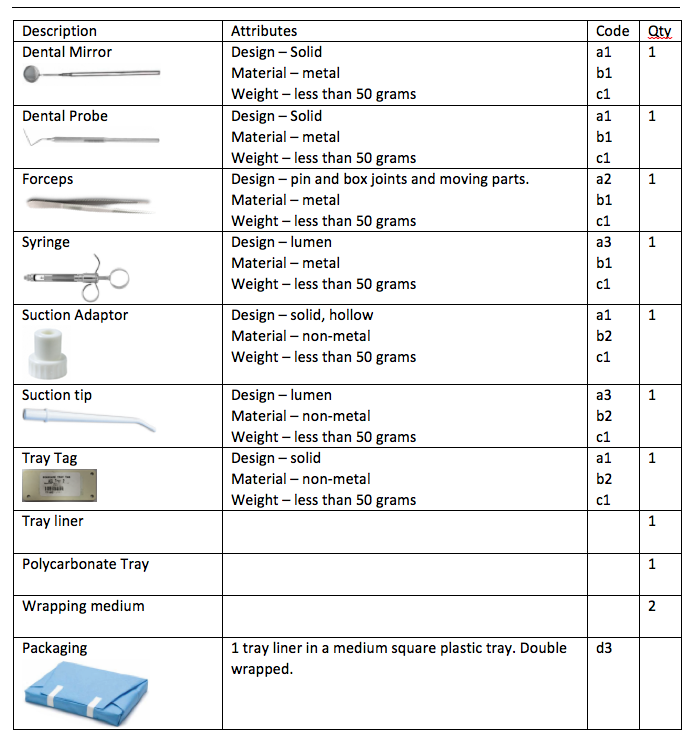
From the table above we can deduce an overall description: low weight metal and non-metal instruments supported on a Tray layer in a small perforated plastic tray. Double wrapped.
Materials used comprise both metal and plastic. The dental tray is made from polycarbonate, which has a low thermal conductivity and therefore presents the greatest challenge.
Dental Tray Configuration Matrix
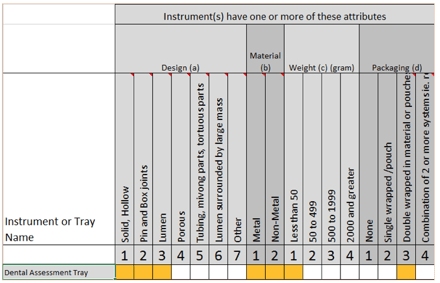 To the right you can see how it looks in our free downloadable matrix:
To the right you can see how it looks in our free downloadable matrix:
Given that we need to select the attributes that pose the greatest challenge to sterilization, the outcome would look like this:
Design (a3)
Material (b2)
Weight (c1)
Packaging (d3)
OK, so this is where you are going to need a copy of ISO/TS 17665-3:2013 - Sterilization of health care products - Moist heat - Part 3: Guidance on the designation of a medical device to a product family and processing category for steam sterilization.
Table 6 in the standard provides a matrix which allows you to identify the Product Family and Steam Penetration Resistance. Unfortunately I can’t reproduce the table due to copyright, but I can however show you the results.
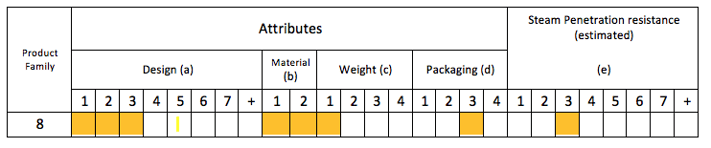
So our Dental Assessment Tray has a Steam Penetration Resistance of (e3) and belongs to Product Family #8.
Conclusion
Once you get the hang of it, the surgical instrument categorisation process is straight forward. With the help of the matrix and the examples here and in the standard, you should be able to work it out.
Just remember that the categorisation process is quite subjective and one person may categorise instruments and trays differently to another.
While AS/NZS 4187 may not provide guidance on product families it does state that the method used to classify an RMD into a product family shall be based on similar attributes to those discussed above and should be documented.
It also states that reference should be made to the manufacturer’s instructions.