What are the minimum label requirements under AS4187?
 Re-usable Medical Devices must, in accordance with AS4187 2014, be labelled prior to sterilisation. The label must identify the contents and provide information for batch control.
Re-usable Medical Devices must, in accordance with AS4187 2014, be labelled prior to sterilisation. The label must identify the contents and provide information for batch control.
But while the standard's requirements are critical, there are also a number of other requirements such as OH&S that may affect the way you label your RMD’s.
AS/NZS4187 minimum label requirements
The standard's reference to the provision of information for batch control would suggest that, at a minimum, the steriliser and cycle number identification must also be provided.
The standard states this as follows:
‘Labelling of a SBS can be achieved by the use of a pre-printed, adhesive label or by writing on the sterilizing indicator tape for a wrapped RMD or on the clear plastic film side of a PSBS outside any sealed section.
If you’re using a gun and sticker manual system, you will probably meet this requirement by producing a sticker that has the sterilizer number and cycle number and placing it on the pack. You then probably write the name of the contents on the sterilizer tape.
The question is whether this is enough information to achieve effective batch control. For example, what about expiry? Without a date, how will you know if your pack has expired? You could probably "solve" his issue by creating either another sticker or by writing the date on the sterilizer tape.
If you look at the standard you will discover that there are 2 criteria that define a non-conforming product:
- An RMD that has exceeded its expiry date
- An RMD that has a missing or defective batch control label.
The need to identify who sterilized what
 Faced with the requirement to track an RMD for recall, the process will benefit from knowing who was responsible for packing and sterilizing the item in question. What are the implications of not having this information readilly available when dealing with an already stressful incident?
Faced with the requirement to track an RMD for recall, the process will benefit from knowing who was responsible for packing and sterilizing the item in question. What are the implications of not having this information readilly available when dealing with an already stressful incident?
Providing this supplemental information in a manual fashion is inefficient and subject to human error.
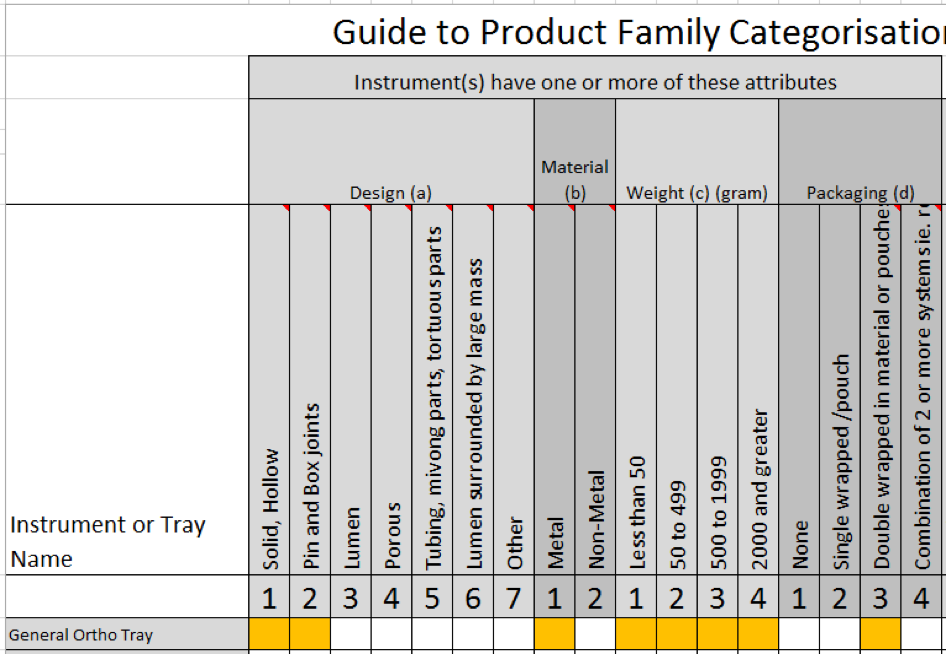
But that's not all because changes introduced to the standard in 2014 are soon to be compulsory. I am talking about product families.
AS4187 Product Families coming into play
Product families are critical to defining the validation and re-qualification of cleaning, disinfecting and sterilizing process. The goal is to ensure that each RMD can be safely and effectively processed.
Unless you work in a very small Health Service Organisation (HSO) and process only one type or one product family of RMD’s, you’ll need a way to identify which RMD’s belong to which product family. You will also need to guarantee that an RMD of one product family does not get sterilized in a cycle type designed for another product family. The danger of mixing up cycling types is that the functional integrity of the RMD may be comprised and the RMD may not achieve the required parameters to be effectively sterilized.
For this reason it is logical that either the name of the product family or the suitable cycle type should also form part of the RMD label requirements.
Locating your surgical assets during recall events
In relation to product recall, the standard states:
‘The expectation is that timely recall of a released RMD is necessary where there is evidence of failure during the cleaning, disinfecting, packaging or sterilizing processes for that RMD. Recall of a released RMD can also be necessary where problems have been encountered during transport and storage of a reprocessed RMD’.
Knowing the exact storage location of your RMD’s is essential for a timely recall of RMD’s. Once you have determined which RMD’s are affected and need to be recalled, you need to be able to quickly locate them. But what if the affected RMD’s are not in their allocated storage location? Time spent hunting around storerooms to locate the affected RMD’s is detrimental to recall time.
So how do your staff know where to store the RMD’s? If you’ve only got one storeroom then I guess it’s not a problem. But if you have multiple store rooms your job would be made a lot easier if that information was displayed as part of the labelling requirements. The label would tell them where the item should be stored so they dont put it in the wrong storeroom.
It would be reasonable to expect, that as the standard evolves, so too will the labelling requirements.
Labels that are designed for efficient sterilisation and recall processes
So summing up so far we have a need for the following data on the label:
- Name of the RMD
- Sterilizer ID
- Cycle Number
- Date of Sterilization
- Expiry Date
- Staff ID Packaging
- Staff ID Sterilizing
- Product Family
- Cycle Type
- Storage Location
While these identifiers are not explicitly expressed as requirements in the standard, one could argue that they really are critical to effective management of sterilizing processes.
Finally, there are a number of other important points of identification for RMD’s that, while not relevant to AS 4187, are a requirement under other standards and statutes. For example, for those who process on behalf of clients the packaging must be labelled with the manufacturer and date of manufacture. Additionally, there are OH&S requirements related to lifting that require you to specify the weight of the package on the RMD.
As you can see, there's much to consider regarding minimum product labelling requirements. Especially if you running a manual system. One of the great benefits of using an automated surgical tracking system is that is AS 4187 and OH&S compliant. This means that your labels meet all the requirements right out of the box.


![Quick Guide to assembling a dental tray [AS4187 product families]](https://blog.scancare.com.au/hubfs/Tables,_Charts_forms/dental_tray_assembly_table.png)